The FOXJ3 gene helps connect the development of the brain and the condition of epilepsy, which affects more than 50 million people worldwide. Focal cortical dysplasia is the main cause of drug-resistant epilepsy. Researchers have identified that the FOXJ3 gene is the master regulator of the PTEN-mTOR pathway, which is a critical pathway for the growth, division, metabolism, and survival of all body cells. When disease-causing FOXJ3 variants are present, they cannot turn on the PTEN tumor suppressor. This over-activation of the mTOR pathway results in the formation of unusually large and irregularly shaped neurons, which is the main cause of focal cortical dysplasia. This newly discovered role of the FOXJ3 gene provides new insight into mTOR pathway-related diseases and the possibility of developing new treatments and diagnostic techniques for people with epilepsy.
FOXJ3 Gene: A Master Regulator of Brain Development
Image Source: Medical Xpress
What FOXJ3 Does in Brain Development
The FOXJ3 gene is a member of the Forkhead Box family of transcription factors, which are proteins that regulate the activity of genes and are critical for development. FOXJ3 is mainly involved in the management of the development of neurons in the early stages of the formation of the brain. Research has identified that FOXJ3 is very active in the neural progenitor cells that produce neurons during the early stages of the formation of the brain. These progenitor cells are present in specific parts of the brain where the formation of neurons is occurring.
The FOXJ3 gene is active in a very specific and timely fashion. FOXJ3 is very active in the progenitor cells in the ventricular zone and the subventricular zone of the developing cortex in both mice and humans. FOXJ3 is significantly reduced in activity after embryonic day 15.5.
The Impact of Mutations in FOXJ3 on the Structure of the Layers of the Cortex
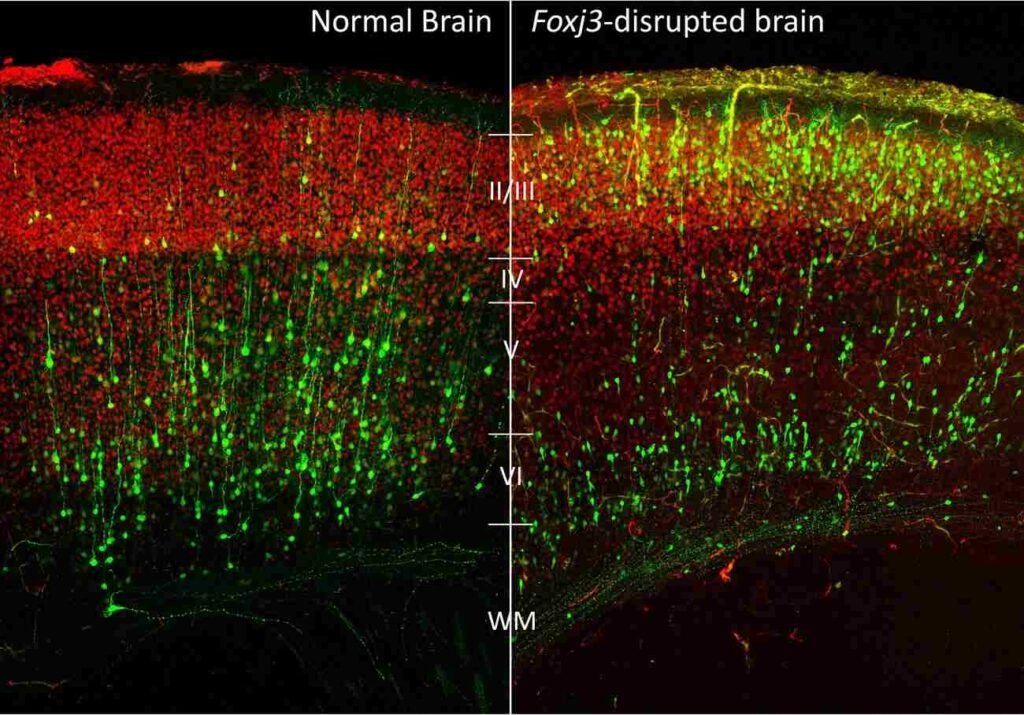
The formation of the cerebral cortex has six layers. Neurons develop in the cortex in a particular pattern from the inside out. Mutations in the function of the FOXJ3 gene mean that the neurons develop in the wrong layers. Reducing the function of the FOXJ3 gene at embryonic day 12.5 and 13.75 results in the continued division of cells and the slowing of neurogenesis. At embryonic day 15.5 and 16.75, the migration of the neurons slows down, and the cell fate of the neurons changes at day 7 after birth when the function of the FOXJ3 gene is reduced.
The effect of the reduced function of the FOXJ3 gene is the formation of larger and abnormal-sized neurons. Such abnormal neurons are characteristic of the focal cortical dysplasia that occurs in the brain of individuals. The function of the FOXJ3 gene determines the depth and the upper layers of the neurons. Without the proper function of the FOXJ3 gene, the structure of the brain becomes abnormal. Such abnormalities in the structure of the brain create problems that lead to epileptic seizures.
The Relationship between the PTEN-mTOR Pathway and the Function of the FOXJ3 Gene
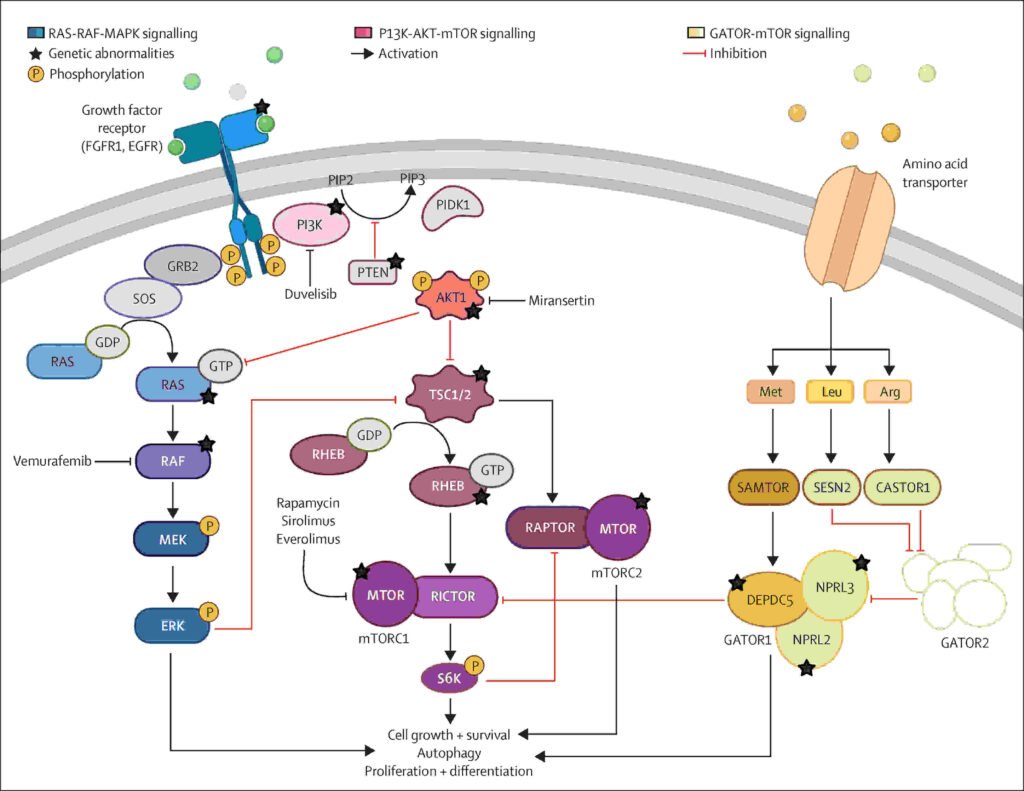
The PTEN gene is a significant regulator of the mTOR pathway. Studies have demonstrated that the PTEN gene is directly controlled by the FOXJ3 gene. A disease-causing mutation in the FOXJ3 gene in focal cortical dysplasia type II leads to the formation of the p.N351S mutant. This mutant has lost the ability to activate the PTEN and TSC1 genes. The reduction of the function of the FOXJ3 gene leads to the reduction of the PTEN function and the activation of the pS6 pathway. PTEN activation can reverse the cortical abnormalities that occur in the brain.
Understanding How FOXJ3 Helps in the Migration and Positioning of Neurons
The Neural Progenitor Cells and the Function of the FOXJ3 Gene
The function of the FOXJ3 gene in the cortex of the developing mouse brain has a particular temporal and spatial pattern. Protein tests carried out from embryonic day 11.5 to 18.5 show that the level of the protein in the cortical plate increases. High levels of the protein occur from embryonic day 12.5 to 18.5. However, the level of the protein in the ventricular zone decreases significantly from embryonic day 12.5 to 14.5. This halt in the developmental process is an important checkpoint. Neural progenitor cells now shift from making deep layer neurons to making upper layer neurons.
Why Neurons End Up in the Wrong Cortical Layers
Experiments involving the knockdown of the gene FOXJ3 reveal exactly why neurons end up in the wrong place. It has been observed that if the gene is knocked down by RNA interference at embryonic day 14.5, it results in significant delays in migration. While 53.1% of the cells actually end up in the cortical plate, 38.0% end up in the intermediate zone. Neurons that were electroporated at E12.5 end up in the outer Layer 2/3 rather than the deeper Layer 5/6. Layer-specific marker studies reveal that these neurons express SATB2 but not CTIP2, suggesting that even in the wrong place, these neurons still end up as callosal projection neurons.
FOXJ3’s Role in the Six Layered Cerebral Cortex Development
Normally, the six layers of the cerebral cortex contain neurons that develop in an inside-out manner. However, if the gene is not functioning properly, it results in the normal developmental architecture not being followed. Neurons end up as callosal projection neurons irrespective of their birth time. Neurons born at E13.75 that normally end up in Layer 4 end up with lower RORB expression, only 9.5% as opposed to 80% in the control. FOXJ3 regulates genes that control transcription and the cell cycle in progenitors.
Disease-Associated Variants and Their Effects on PTEN Activation
The FCD-associated variant “p.N351S” fails to activate PTEN expression at all. This causes the mTOR pathway to remain active, which results in the large cell bodies observed in Focal Cortical Dysplasia II. When PTEN expression is restored in the experiments, the cortical abnormalities are corrected, thus affirming that the FOXJ3 PTEN pathway is the primary pathway.
International Research Breakthrough Connects Genetics to Clinical Cases
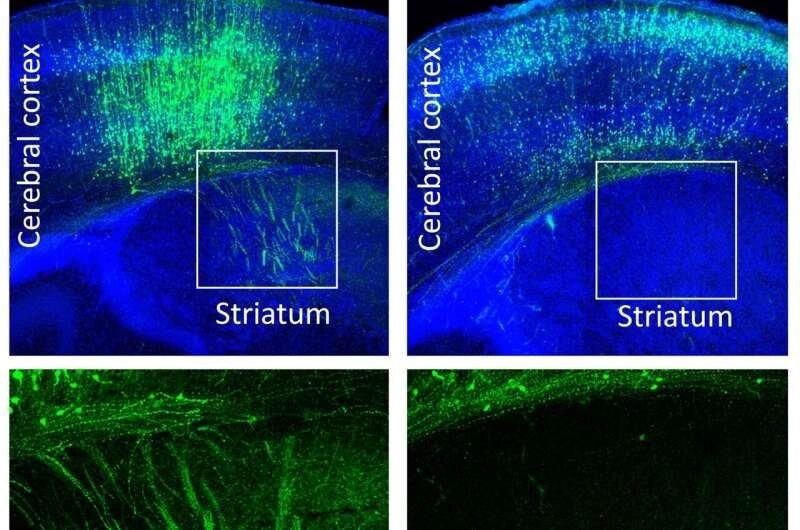
Image Source: Medical Xpress
How Taiwan and UK Teams Found the Genetic Link
Scientists from Taipei Veterans General Hospital and the University College London Institute of Neurology collaborated to identify the genetic link. The first patient had focal seizures that began when the patient was 6 years old. MRI scans indicated that the patient had Focal Cortical Dysplasia in the right frontal gyrus. Since the epilepsy was not controlled with drugs, the patient had surgery to remove the affected brain area. Tests confirmed that the patient had Focal Cortical Dysplasia IIa. Most importantly, the patient’s mother and cousin also had focal epilepsy with possible FCD on imaging.
Families with Drug-Resistant Epilepsy Studied
The researchers conducted whole-genome sequencing on the index case, the cousin, and the asymptomatic grandmother. The heterozygous FOXJ3 variant (c.1052 A>G, p.Asn351Ser) was identified in the affected members of the family. The variant was extremely rare, at 0.000008/2 in TOPMED, 0.00002/5 in GnomAD_exomes, and 0.0003 in the Taiwan View database.
Single-Cell Analysis Reveals Developmental Disruptions
The researchers conducted the study on 170 patients with malformations of cortical development and 76 patients with epilepsy at Taipei Veterans General Hospital. The study was conducted not only on the population in Taiwan but also on the population in the United Kingdom using 85,000 whole-genome sequencing datasets from the Genomics England database and 30,000 whole-exome sequencing datasets from the UCL Institute of Neurology patients. The researchers identified two other heterozygous FOXJ3 missense variants (p.Ile621Val and p.Pro253Thr) in unrelated individuals with focal epilepsy.
Clinical Implications for Epilepsy Diagnosis and Treatment
Improving Genetic Testing for Focal Cortical Dysplasia Patients
For genome sequencing, the results provide a diagnosis 48 percent of the time, for exome sequencing 24 percent, and for multi-gene panels 19 percent. The American Epilepsy Society suggests that genome sequencing and exome sequencing should be considered first-tier tests for patients with unexplained epilepsy. Finding the FOXJ3 mutation opens the door to more possibilities for patients with epilepsy, especially for patients with normal brain MRI scans who had no previous genetic diagnosis.
Targeting mTOR Pathway Opens New Treatment Possibilities
Results from mTOR inhibitors prove to be effective. Everolimus was effective for 40 percent of patients with high drug exposure. This inhibitor had a median seizure reduction of 39.6 percent. Since FOXJ3 is an upstream regulator in the PTEN-mTOR pathway, this treatment is more targeted.
What This Means for the 50 Million Epilepsy Patients Worldwide
Epilepsy patients worldwide will benefit from the fact that a third of all epilepsy patients do not respond to the current drug regimen. Genetic diagnosis will help in the treatment of patients.
Relief and Guidance for Families Seeking Answers
Receiving a genetic diagnosis brings closure to families. It also brings guidance for family planning. In addition, it will help patients qualify for a clinical trial. This diagnosis will help alleviate guilt for the parents.
Gene-Based Therapies on the Way
In the lab, gene therapy reduced seizures by 87 percent in mice with the human form of focal cortical dysplasia. Human tests are expected in about five years.
Conclusion
Epilepsy caused by the FOXJ3 gene is changing the face of research into the disease. This is because it has been able to establish that brain malformations cause seizure disorders. This has helped doctors in the diagnosis and treatment of the disease. It has also helped families find answers in the midst of an untreatable disease with medicine. As gene therapy is being developed, people suffering from focal cortical dysplasia have hope in their future.
FAQs
Q1. How does epilepsy affect a child’s brain development?
Epilepsy does not only affect the brain of an affected child. Epilepsy has been known to affect brain development in children in ways that do not involve seizures. It has been known to affect brain development even if seizures are well controlled. Epilepsy has been known to affect brain development in terms of thinking skills such as language, memory, and attention. Thinking skills are important in the growth of a child.
Q2. Is epilepsy inherited from parents?
Epilepsy is caused by genetics in 30 to 40 percent of people suffering from it. This implies that if there is a genetic component in the causation of the disease, parents, siblings, and children of an affected individual have a two- to four-fold risk of suffering from it.
Q3. Can people with genetic epilepsy become seizure-free?
The outcome of epilepsy in people is quite variable. Some people live with the condition throughout their lives, while others may live with the condition for only a short time. In addition, the condition may disappear in certain people, depending on the cause and the outcome of the treatment.
Q4. What is the role of the FOXJ3 gene in the causation of epilepsy?
FOXJ3 is a master regulatory gene in the causation of epilepsy. The FOXJ3 gene controls the formation of neurons and the placement of these neurons in the right locations in the six-layer cortex of the brain. FOXJ3 mutations result in abnormal neuronal formation and placement in the wrong locations in the brain. This abnormality in the brain is referred to as focal cortical dysplasia and is a major cause of drug-resistant epilepsy.
Q5. How can genetic testing help the patients and the families of people with epilepsy?
Genetic testing can provide a diagnosis for the condition in as many as 48 percent of patients with epilepsy and is therefore recommended as the first diagnostic test for people with unexplained epilepsy. A genetic diagnosis can provide the families of people with epilepsy with the answers they need, end the diagnostic journey for the affected persons and their families, and provide the affected persons with access to targeted treatments and clinical trials of new therapies.
Read My Website Recent News Article: New Study Reveals